Objective, Timely and Reliable Quality Control
Embryotools is an external and independent laboratory that provides quality control (QC) services specifically designed to detect toxicity in all types of materials, culture media, products or equipment used routinely in IVF laboratories. Our QC tests are designed by scientists, doctors, with more than 30 years of experience with both human and mouse gametes and embryos, giving manufacturers, suppliers and end-users maximum confidence about the services provided.
Our state-of-the-art facilities, accredited with the ISO 17025, operate under strict GLP standards 7 days a week and are annually audited by the Spanish Accreditation Body, ENAC. Our flexibility, together with our experience, allow us to provide accurate information, detailed reports and certificates of analysis in the shortest period of time (within 7-10 working days from test article receipt). In addition, we have developed a customized an online platform to manage all QC assays, which allows our customers to easily place their orders and follow the status of their shipments and requests and download their certificates at anytime and from anywhere.
Embryotools’ scientific team has been directly involved in the daily management of IVF Laboratories and therefore understands the specific quality control needs for each material and product used in assisted reproduction procedures. The test results will help ensure that the products can be used in the IVF laboratory with full confidence and peace of mind that they are safe for human gametes and embryos.
Quality control tests for screening of toxicity
Mouse Embryo Assay (MEA)
Our mouse embryo assays (MEA) are ISO 17025 accredited and performed with one-cell stage embryos from an hybrid strain to ensure the highest sensitivity, consistency and reliability of results. We offer different levels of tests that have been optimized to increase the chances of detecting the eventual presence of toxicity on each type of product. The release criteria of our MEAs exceed the standards requested by regulatory agencies to ensure the assays meet the standards expected by our customers and are not simply a regulatory requirement for batch release.
We offer the following MEA options:
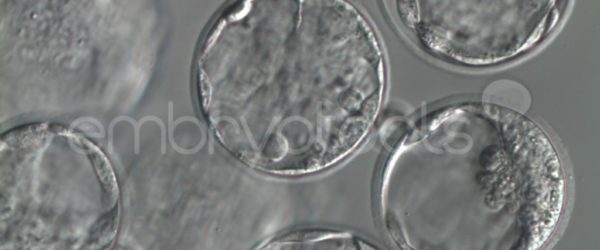
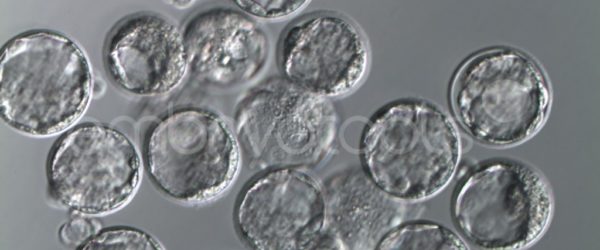
- MEA Standard: mouse embryos are exposed to the test product (through an appropriate protocol, depending on the type of product) and are cultured in vitro for 5-6 days. At least 80% of the embryos must reach the expanded blastocyst stage and pass a strict daily visual examination of morphological quality.
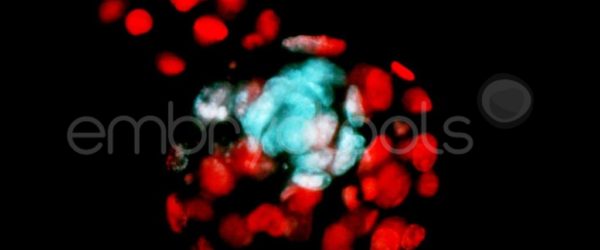
- MEA Standard plus (with cell counts): after performing a standard MEA, the blastocysts obtained are subjected to a staining protocol to count the total number of cells, which allows to ensure the maximum accuracy on the evaluation of the embryo quality.
- MEA with time-lapse (MET): In this variation of the standard MEA, the development of the embryos is monitored in time-lapse incubators, which provides information on the morphokinetic data of all the cell divisions and stages of the embryo.
- MEA with time-lapse and cell counts (MET with cell counts): after performing a MET, the blastocysts obtained are subjected to a staining protocol to count the total number of cells, which allows to ensure the maximum accuracy on the evaluation of the embryo quality.
- MEA with in vitro fertilization (MEA FIV): Mouse gametes are exposed to the test product before proceeding with in vitro fertilization. Properly fertilized oocytes are cultured for 6 days. As in the standard MEA, a minimum of 80% of all embryos in culture must develop to the expanded blastocyst stage and pass a strict daily visual examination of morphological quality.
Other customized MEAs can also be offered upon request.
Limulus Amebocyte Lysate Assay (LAL)
The Limulus Amebocyte Lysate assay (LAL) is the most sensitive and specific test currently available to detect the presence of endotoxins in all types of materials, culture media or products used in the IVF laboratory. All protocols have been carefully optimized and validated for each type of product and are currently part of the schedule of our ISO17025 accredited tests.
Human Sperm Motility Assay (SMA)
The human sperm motility assay (SMA) is also ISO 17025 accredited and usually used as a quality control tool to test all types of devices or materials that are usually used in the processing of semen in IVF laboratories.
For more information about our QC tests and services, please contact us at: info@embryotools.com